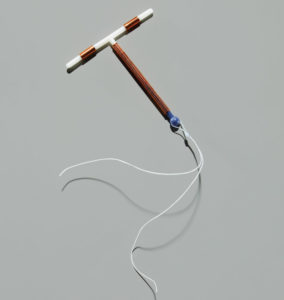
 The ParaGard T-380A IUD Birth Control Device has been quite popular. Manufactured by Teva Pharmaceuticals until the fall of 2017, and by CooperSurgical since that time, it is a small, T-shaped device that uses copper to prevent pregnancy as opposed to hormones; the only hormone-free IUD currently on the market. However, “arm-breakage” issues, particularly during removal of the device, have caused thousands of women to undergo surgery to remove the broken arm(s) and repair any associated damage caused by them.
The ParaGard T-380A IUD Birth Control Device has been quite popular. Manufactured by Teva Pharmaceuticals until the fall of 2017, and by CooperSurgical since that time, it is a small, T-shaped device that uses copper to prevent pregnancy as opposed to hormones; the only hormone-free IUD currently on the market. However, “arm-breakage” issues, particularly during removal of the device, have caused thousands of women to undergo surgery to remove the broken arm(s) and repair any associated damage caused by them.

If you were implanted with a ParaGard T-380A IUD and required surgery due to an “arm breakage”, contact us online or call us at 1-866-252-3535.
